http://www.ncbi.nlm.nih.gov/pubmed/23248118
Abstract
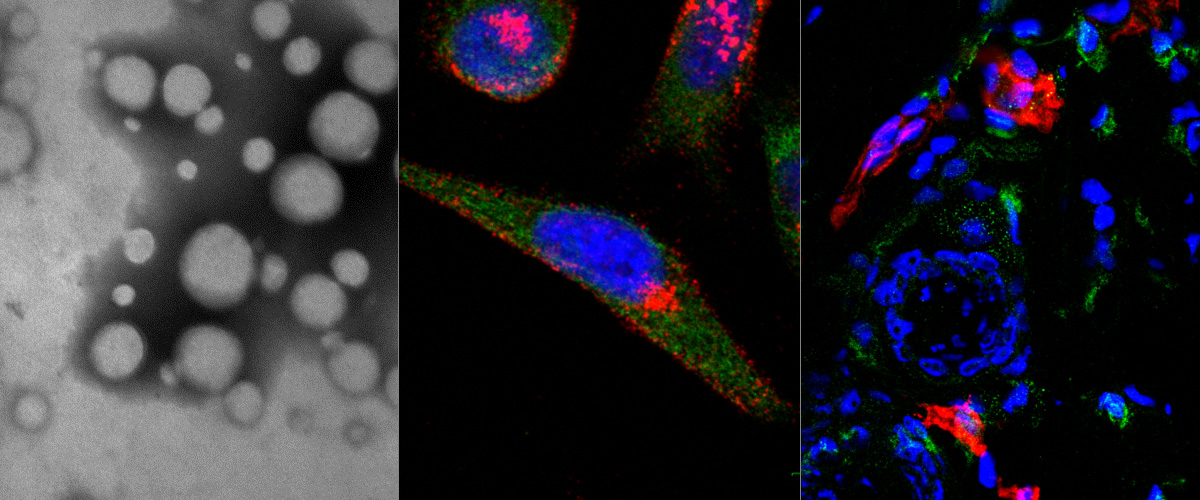
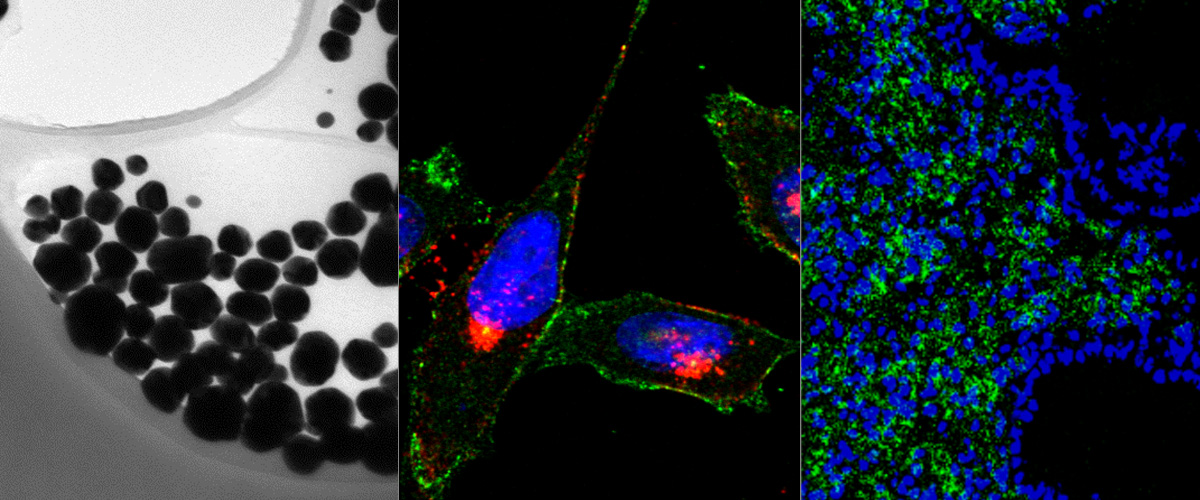
Bit1 is a proapoptotic mitochondrial protein associated with anoikis. Upon cell detachment, Bit1 is released into the cytoplasm and triggers caspase-independent cell death. Bit1 consists of 179 amino acids; for the C-terminal, two thirds of the molecule functions as a peptidyl-tRNA hydrolase, whereas the N-terminus contains a mitochondrial localization signal. Here, we localize the cell death domain (CDD) to the N-terminal 62 amino acids of Bit1 by transfecting cells with truncated Bit1 cDNA constructs. CDD was more potent in killing cells than the full-length Bit1 protein when equivalent amounts of cDNA were transfected. To develop Bit1 CDD into a cancer therapeutic, we engineered a recombinant protein consisting of the CDD fused to iRGD, which is a tumor-specific peptide with unique tumor-penetrating and cell-internalizing properties. iRGD-CDD internalized into cultured tumor cells through a neuropilin-1-activated pathway and triggered cell death. Importantly, iRGD-CDD spread extensively within the tumor when injected intratumorally into orthotopically implanted breast tumors in mice. Repeated treatment with iRGD-CDD strongly inhibited tumor growth, resulting in an average reduction of 77% in tumor volume and eradication of some tumors. The caspase independence of Bit1-induced cell death makes CDD a potentially attractive anticancer agent, because tumor resistance to the main mechanisms of apoptosis is circumvented. Using iRGD to facilitate the spreading of a therapeutic agent throughout the tumor mass may be a useful adjunct to local therapy for tumors that are surgically inoperable or difficult to treat systemically. Copyright © 2012 Elsevier Inc. All rights reserved.